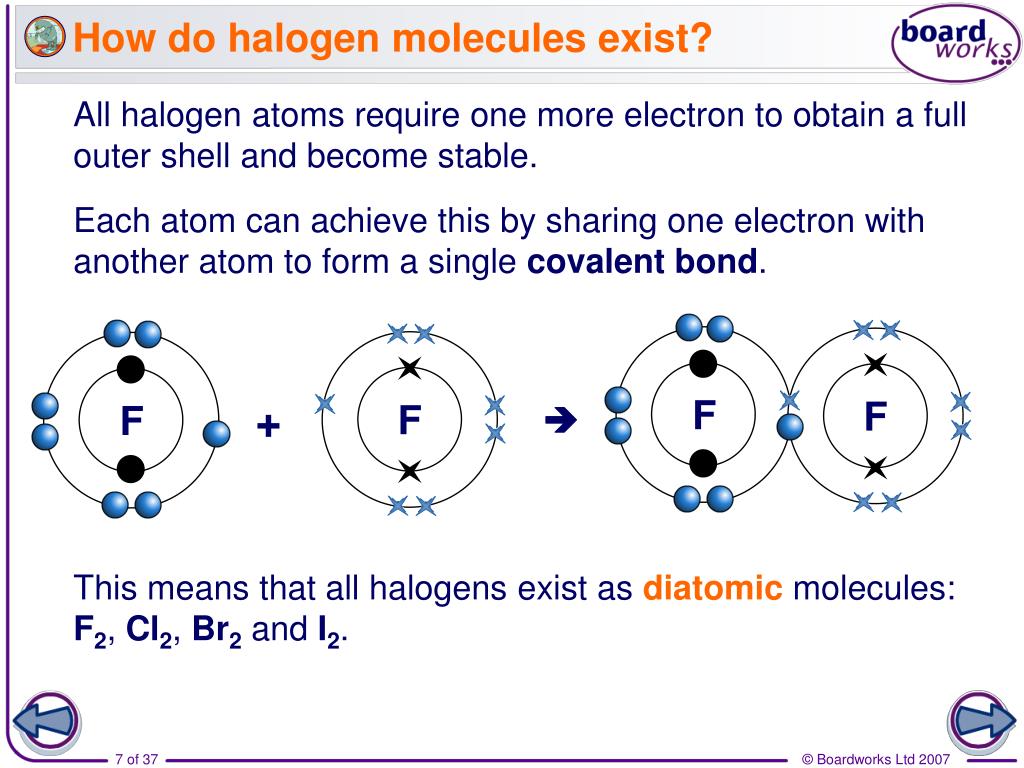
Therefore, the non-metallic properties decrease from fluorine to Iodine. (I.E.) value decreases with Increase in size of atom i.e. They have no tendency to lose the electron.Īmong halogens, the I.P. Since atomic radii of halogens are smallest in their respective period, their ionization potentials are very high. The energy required to remove outermost electron from the gaseous atom of an element, when it is in the ground state is called ionization potential or ionization enthalpy. Ionization Potential (I.P.) or Ionization Enthalpy: Hence fluorine has only –1 oxidation state. It does not have ‘d’ orbitals in Its valence shell. Fluorine has Only -1 Oxidation State:įluorine is the most electronegative element having highest electronegativity 4. Therefore, they can show positive oxidation states like +1, +3, +5 and +7.įluorine has only -1 oxidation state due to the absence of vacant n-’d’ orbitals. These elements when combining with the more electronegative element, their electrons of nth orbit get promoted to n-‘d’ orbital. Hence they are monovalent.Ĭhlorine, bromine and Iodine have empty n-‘d’ orbital. Therefore, the common valency of halogen family is 1. They attain the octet either by accepting an electron to form a univalent anion, X-, (F-, Cl-, Br- and I-) by sharing the unpaired electron with the unpaired electron of another atom to form a covalent bond (as in Cl2, Br2, HCI, HF etc). They need one electron to complete their octet. They contain seven electrons in the valence shell. 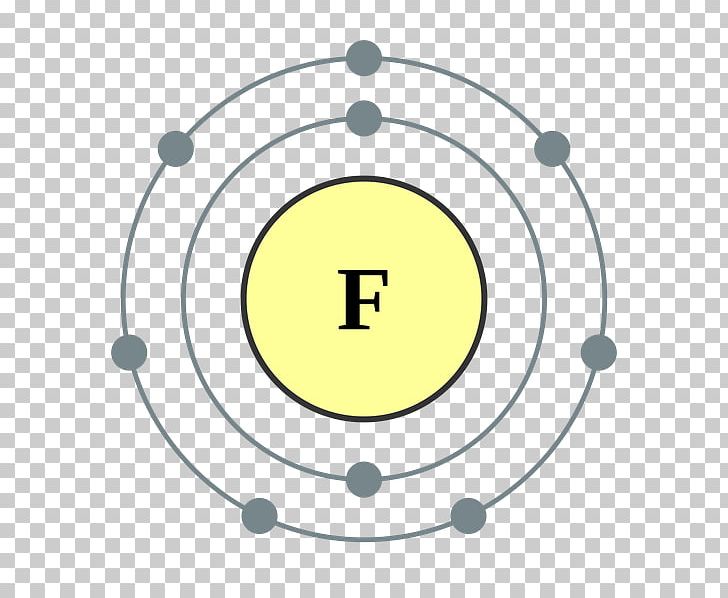
So these elements require only one electron to complete the octet.Īll halogen have shell electronic configuration is ns2 np5. There are seven electrons in the outermost shell.Hence they are kept in group VII-A (17) of a periodic table, before inert gases. All halogens contain seven electrons in their outermost shell.All orbits except the last orbit are completely filled.The last electron during configuration occupies p orbital, hence these elements are p block elements.They have characteristic outer orbit configuration of ns2 np5. Characteristic Electronic Configuration of Halogens:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
